S as function of P and T. Integration of (92) again, for pure phase component yields:
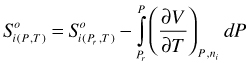
(112)
Expansion in an ideal gas and a deviation term:
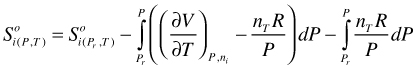
(113)
Solving the last integral in (113):
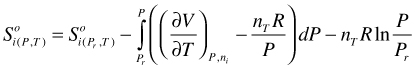
(114)
Extending the lower integration limit:
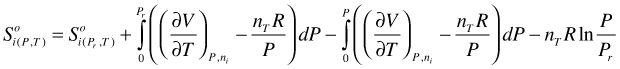
(115)
The first two terms of (110) are the S of a pure hypothetical ideal gas. If the system is a mixture ideal mixing entropy has to be added
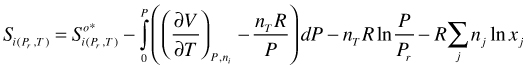
(116)